A new study published in ACS Sensors brings together a long-standing collaboration between Dynamic Biosensors (a Bruker Corporation company), AstraZeneca, TU Dresden, and the University of Dundee, including leading experts such as Alessio Ciulli and Stefan Geschwindner.
This project reflects how collaboration between academia and industry can open new perspectives on complex biological systems, and KSI is proud to have been actively involved.
A new lens on PROTAC mechanisms
PROTACs are molecules designed to bring a target protein and an E3 ligase together, forming a ternary complex that triggers protein degradation. Understanding how this complex forms is key, but difficult to measure with conventional methods.
This study introduces a fluorescence-based approach that can separately track binary and ternary interactions in real time. By distinguishing these states within a single experiment, it provides a clearer and more detailed view of how PROTAC systems behave.
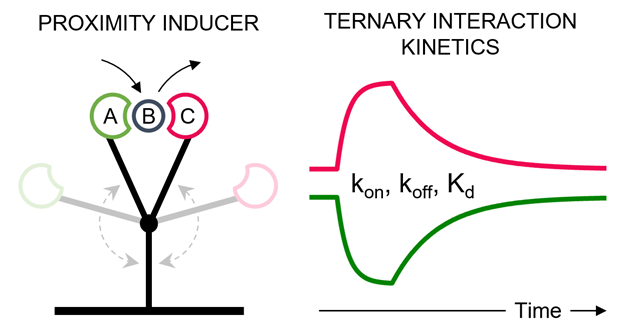
Engineering proximity to reveal kinetics
At the core of the method is a Y-shaped DNA nanostructure that positions proteins in controlled proximity on a biosensor surface. This setup enables direct observation of how ternary complexes form, evolve, and dissociate, revealing kinetic pathways that are often hidden in traditional assays.
The approach is also sensitive to weak or transient interactions and supports high-throughput screening, making it highly relevant for drug discovery applications.
Overall, this work shifts the focus from simply measuring binding strength to understanding the full kinetic and mechanistic landscape of PROTAC function.
KSI is proud to contribute to collaborations that connect academic insight with industrial innovation in drug discovery, and congratulates all scientists that participated on this project!
Read the full publication here.